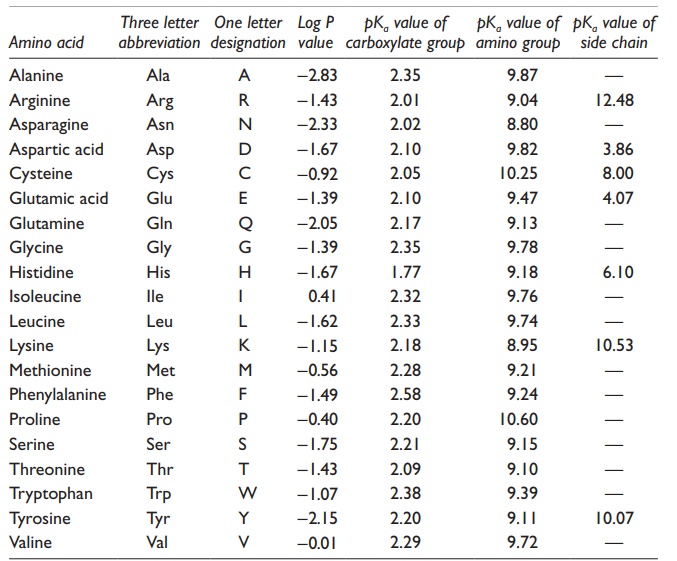
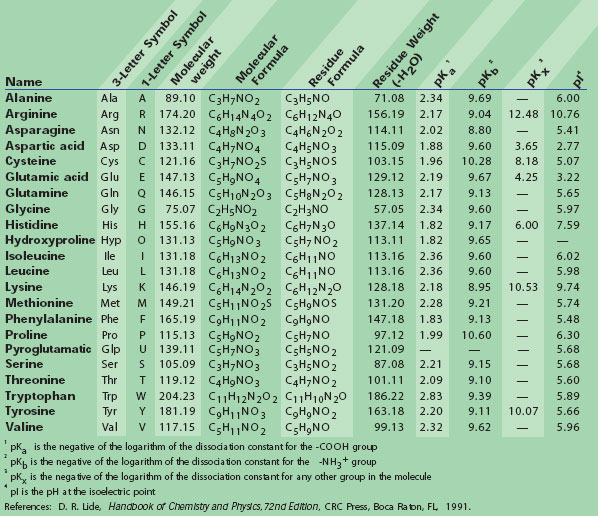
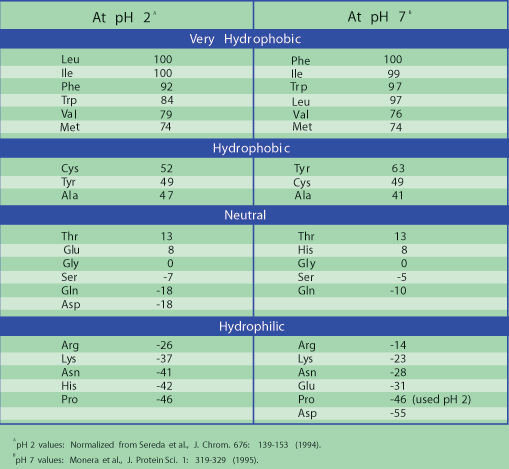
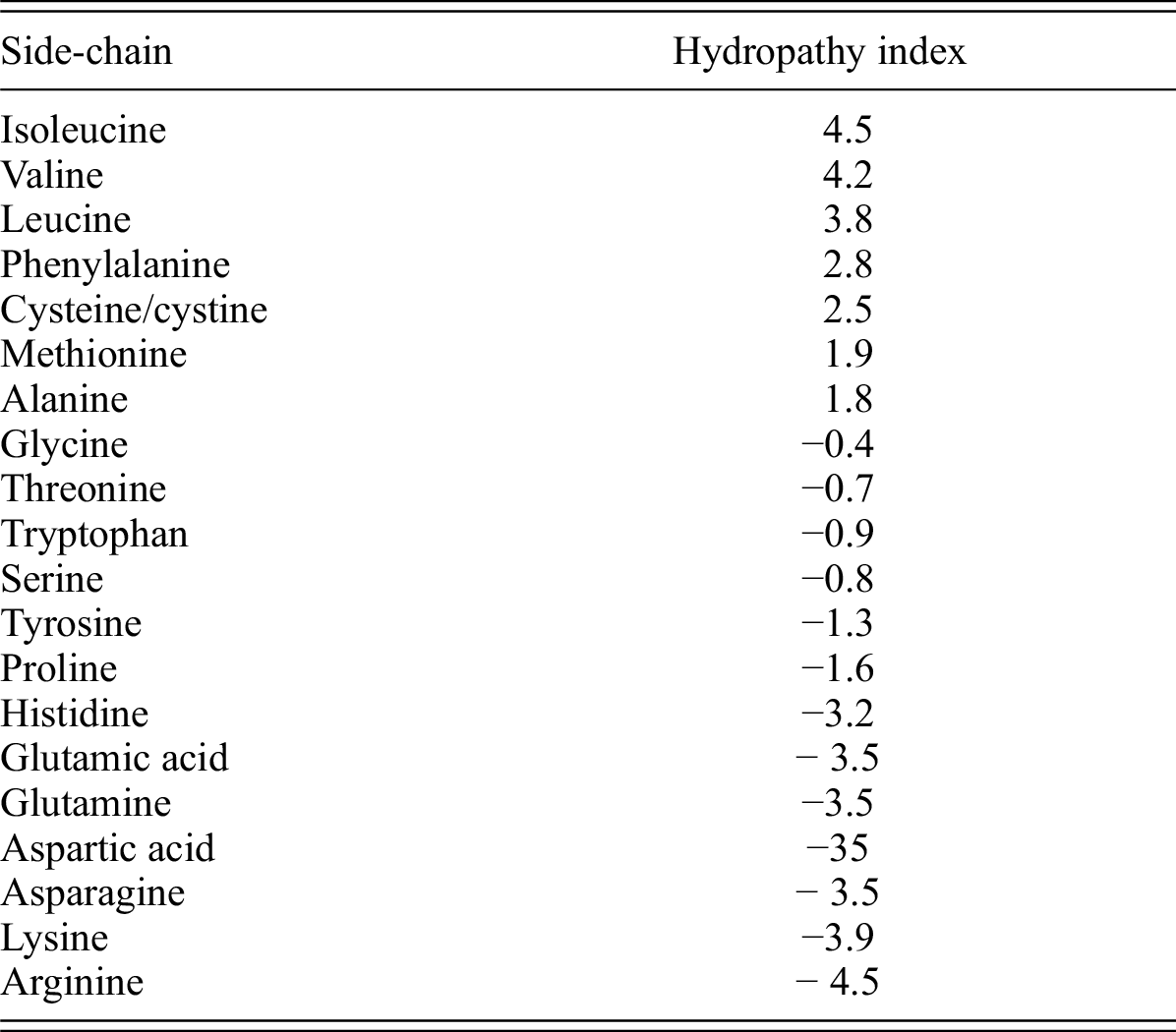
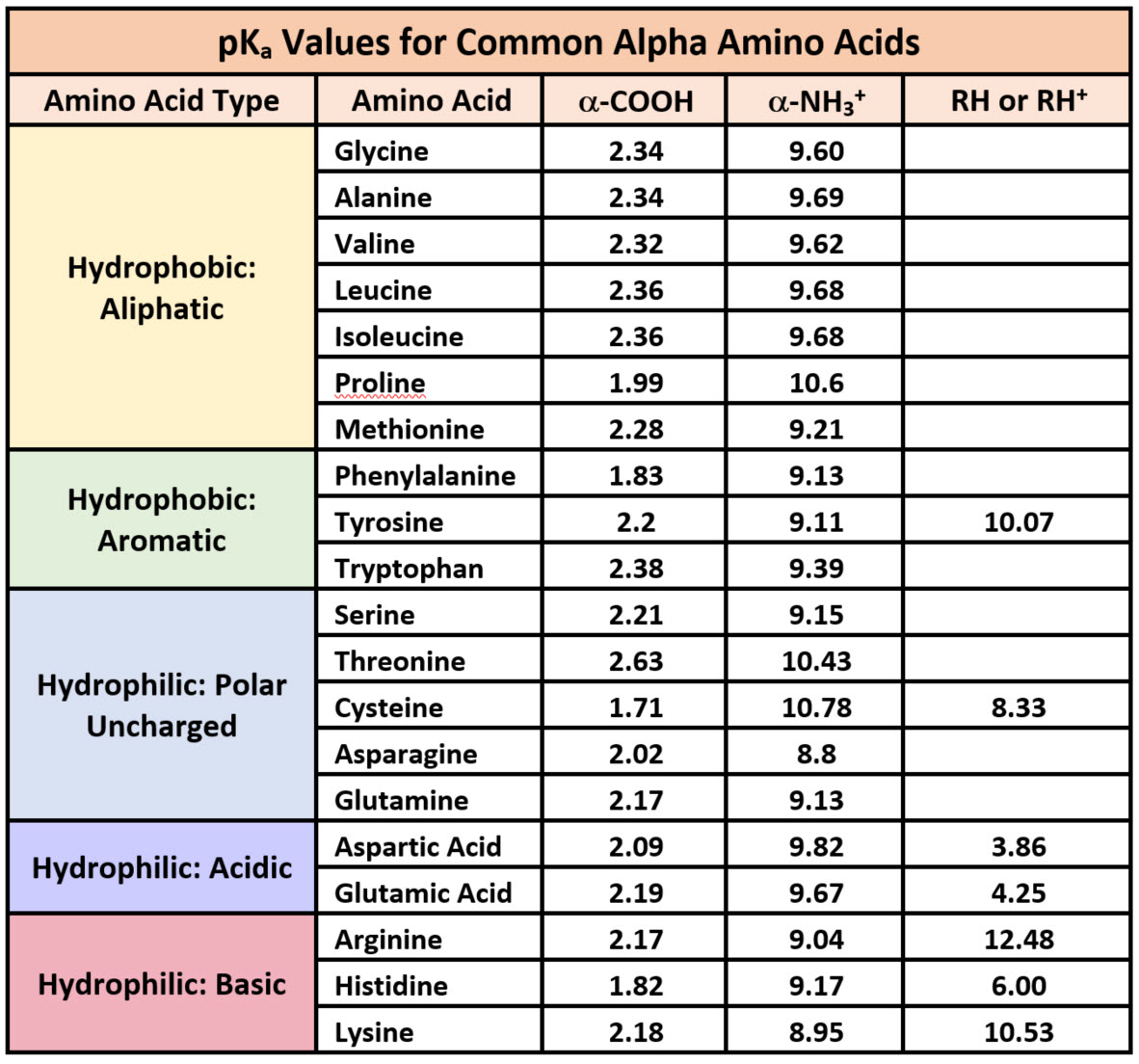
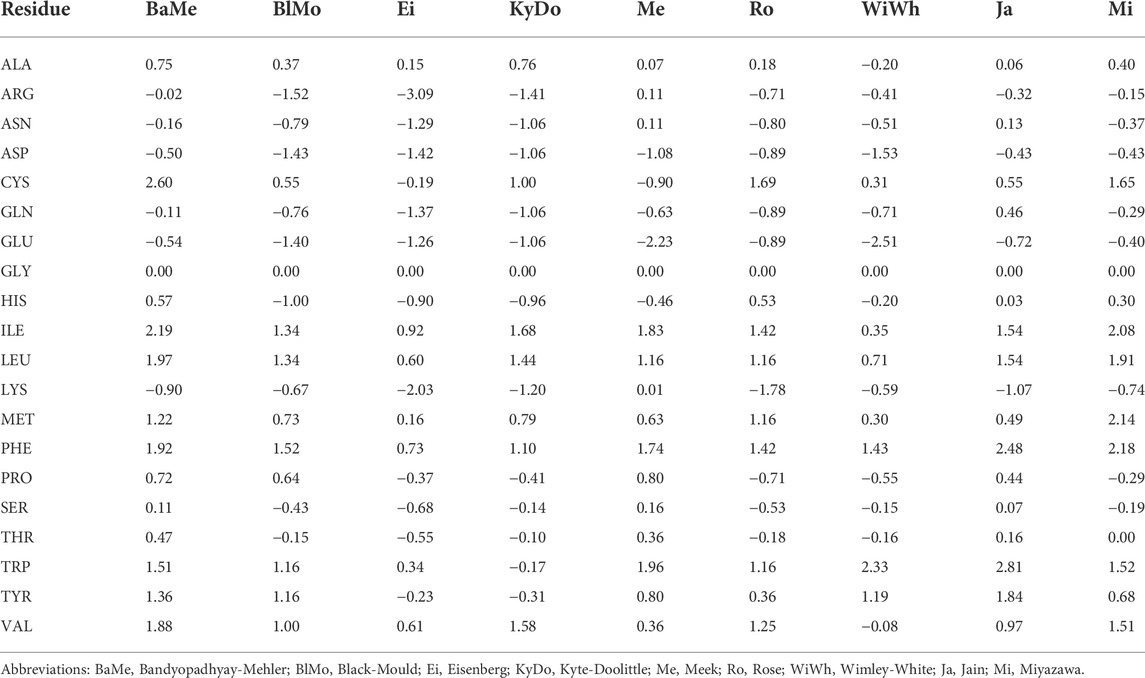
N-Capping Motifs Promote Interaction of Amphipathic Helical Peptides with Hydrophobic Surfaces and Drastically Alter Hydrophobicity Values of Individual Amino Acids | Analytical Chemistry

Proteins: Amino Acid Chains DNA Polymerase from E. coli Standard amino acid backbone: Carboxylic acid group, amino group, the alpha hydrogen and an R group. - ppt download

Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers | PNAS
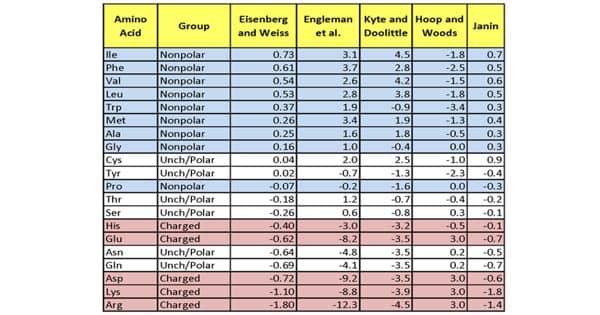
Hydrophobicity Scales - more Hydrophobic Amino Acids have Highest Positive Values - Assignment Point
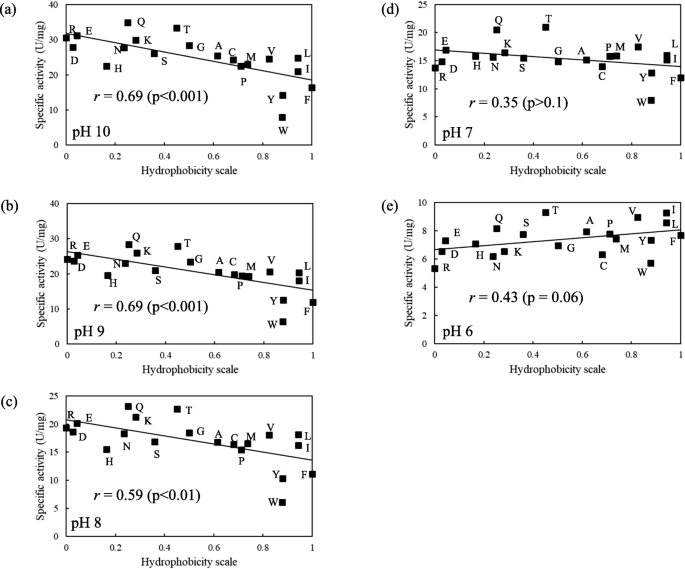
The hydrophobicity of an amino acid residue in a flexible loop of KP-43 protease alters activity toward a macromolecule substrate | Applied Microbiology and Biotechnology

Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers | PNAS

A simple atomic-level hydrophobicity scale reveals protein interfacial structure. | Semantic Scholar